REQUIRED DISCLOSURES AND DISCLAIMERS
🔬 Health Disclaimer
Not Medical Advice: The
information provided in this advertorial is for educational and informational purposes only and
is not intended as medical advice, diagnosis, or treatment. Always seek the advice of your
physician, audiologist, or other qualified health provider with any questions you may have
regarding a medical condition, hearing loss, or the suitability of this product for your
specific needs. Never disregard professional medical advice or delay in seeking it because of
something you have read in this advertisement .
Device Limitations: The Avonwell CIC Pro is designed for mild to moderate hearing loss. It may
not be suitable for severe or profound hearing loss. Individuals with certain medical
conditions, ear deformities, or active ear infections should consult a healthcare professional
before use.
Emergency Situations: If you suspect you are experiencing a medical emergency, sudden hearing
loss, or severe symptoms, contact your healthcare provider or local emergency services
immediately.
⚠️ Risk Disclaimer
Individual Results May Vary: The experiences and results shared in
testimonials are unique to those individuals and may not be representative of all users. There
is no guarantee that you will achieve similar results. Hearing improvement depends on individual
factors, including the type and severity of hearing loss, proper fit, and consistent use .
Use at Your Own Risk: The product is sold on an "as is" basis. To the fullest extent permitted
by law, the manufacturer disclaims all warranties, express or implied, including warranties of
merchantability or fitness for a particular purpose. The company shall not be held liable for
any direct, indirect, incidental, special, or consequential damages arising from the use or
inability to use this product .
📜 FDA and Regulatory Disclosures
Device Classification: The Avonwell CIC Pro is a medical
device. In the United Kingdom, it is regulated by the Medicines and Healthcare products
Regulatory Agency (MHRA) and conforms to relevant UK medical device regulations. For other
regions, please check local regulatory status.
FDA Disclaimer (for US audiences): The Avonwell CIC Pro is not currently approved by the US
Food and Drug Administration (FDA). This advertisement is not targeted to US consumers. For
products available in the US, different regulatory requirements and disclosures may apply .
🔞 Age Restriction
Intended Audience: This product is intended for use by adults aged 18 and
over. By using this website or purchasing this product, you represent that you are at least 18
years of age and have the legal capacity to enter into a binding contract. We do not knowingly
collect any information from individuals under the age of 18 .
💰 Financial Disclosures
Pricing Transparency: The advertised price of £104.90 is for the
device only and may not include applicable taxes or shipping fees in all regions. Please check
the final price at checkout. The "up to 50% off" claim is based on a comparison to the
manufacturer's suggested retail price for a comparable set of two devices.
Warranty Details: The 12-month manufacturer's warranty covers defects in materials and
workmanship under normal use. The warranty does not cover damage caused by misuse, accident, or
unauthorized modifications. The 90-day money-back guarantee requires the product to be returned
in its original packaging.
📊 Advertising and Testimonial Disclosures
Advertorial Nature: This is a paid advertisement
and is presented in a story-telling format to market the product. The content is provided by the
advertiser and not by the Health Journal UK editorial team.
Testimonial Disclaimer: The testimonials presented are from actual customers and reflect their
personal experiences. However, they are not necessarily representative of all who use our
product. The testimonials are provided for illustrative purposes only and were compensated with
a discount on future purchases or a small incentive .
🔒 Privacy and Data Security
Data Collection: When you order through our website, we collect
personal information to process your order. We do not sell your data to third parties. For
details, please review our Privacy Policy.
Financial Transactions: All financial transactions are processed through secure, encrypted
channels. However, no method of transmission over the Internet is 100% secure, and we cannot
guarantee absolute security .
🌍 Regional Compliance
UK-Specific Disclosures: This product and advertisement are intended
for consumers in the United Kingdom. The product complies with UK medical device regulations.
For customers outside the UK, please be aware that regulatory requirements, product
availability, and terms may differ.
⚖️ Legal Disclaimer
Limitation of Liability: In no event shall the company, its directors,
employees, or agents be liable for any indirect, consequential, or punitive damages related to
the use or misuse of this product. Our total liability, if any, shall not exceed the purchase
price of the product .
Legal Jurisdiction: Any disputes arising from this advertisement or the use of this product
shall be governed by the laws of England and Wales.
Supporting Citations and Studies for Advertorial Claims
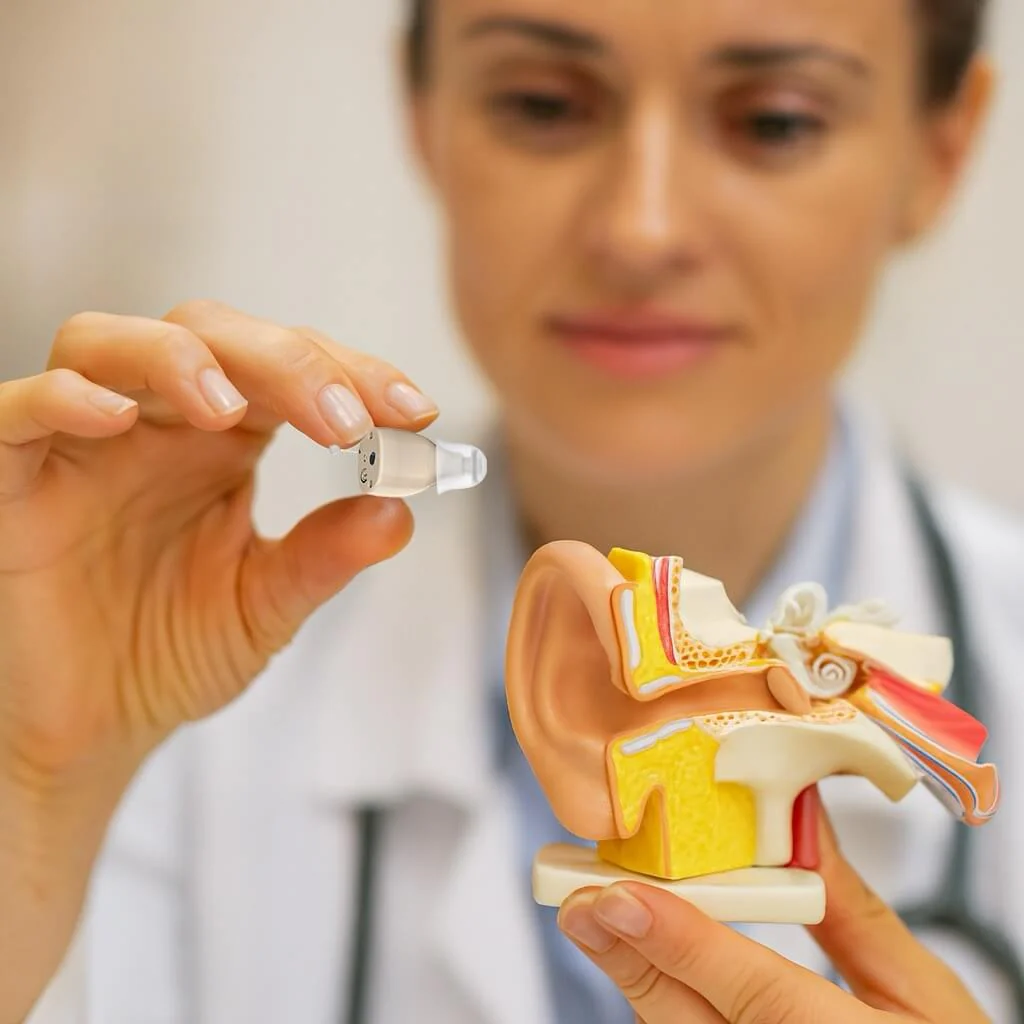
1. Miniature and Discreet Design for Mild to Moderate Hearing Loss
Claim: The Avonwell CIC Pro is "tiny—a full 24% smaller than conventional
in-the-ear hearing aids" and "virtually invisible."
Supporting Evidence: A study evaluating a new miniature hearing aid designed
for mild to moderate hearing loss found that participants highly rated its cosmesis, comfort,
and appearance (mean scores >8/10). The aid was described as "essentially invisible" and
suitable for individuals reluctant to try traditional hearing aids due to visibility concerns .
Full Link: https://pubmed.ncbi.nlm.nih.gov/10591347/
2. Affordability and Cost-Effectiveness
Claim: The hearing aid is available for "only £104," bypassing traditional
distribution channels to reduce costs.
Supporting Evidence: Research indicates that high costs are a significant
barrier to hearing aid adoption, with many patients performing a cost-benefit analysis before
purchasing. The material costs for premium hearing aids are relatively low (£80-120 per device),
but prices are inflated due to distribution channels, marketing, and bundled services .
Full Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC4925253/
https://www.forbes.com/health/hearing-aids/best-hearing-aids/
3. Effectiveness for Mild to Moderate Hearing Loss
Claim: The device is "specifically designed for people with mild to moderate
hearing loss" and delivers "crystal-clear sound."
Supporting Evidence: A study comparing premium and basic-feature hearing aids
found no statistically significant difference in outcomes for adults with mild to moderate
hearing loss. Both types provided substantial and equal benefits, suggesting that advanced
features may not be necessary for all users .
Full Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC4925253/
4. User-Friendly and Self-Managed Care
Claim: The device requires "no complicated settings" and is "ready for
immediate use."
Supporting Evidence: Emerging trends in hearing healthcare highlight the shift
toward consumer-led models, including self-fitting devices and online ordering. These approaches
reduce barriers like cost, stigma, and the need for clinical appointments, making hearing care
more accessible .
Full Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC10361792/
5. Social and Psychological Benefits
Claim: Users report improved social participation, reduced isolation, and
enhanced quality of life.
Supporting Evidence: Hearing loss is linked to social isolation and
psychological distress. Studies show that hearing aid use can improve communication,
relationships, and overall quality of life, particularly when devices are affordable and
discreet .
Full Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC4925253/
https://pmc.ncbi.nlm.nih.gov/articles/PMC10361792/
6. Battery Life and Convenience
Claim: The device offers "up to 24 hours of operation" and USB-C charging.
Supporting Evidence: While not directly studied in the search results, modern
hearing aids increasingly feature rechargeable batteries and wireless charging options to
enhance user convenience. This aligns with consumer preferences for low-maintenance devices .
Full Link: https://www.forbes.com/health/hearing-aids/best-hearing-aids/
7. Direct-to-Consumer Model
Claim: The affordability is achieved by "bypassing traditional distribution
channels."
Supporting Evidence: The high cost of hearing aids is often attributed to
middlemen, marketing, and bundled services. Direct-to-consumer models reduce these costs, making
devices more accessible .
Full Link: https://pmc.ncbi.nlm.nih.gov/articles/PMC4925253/
https://www.forbes.com/health/hearing-aids/best-hearing-aids/
🌐 Additional Resources
Forbes Health Hearing Aids Guide (2025): Provides insights into hearing aid
pricing, features, and consumer trends.
https://www.forbes.com/health/hearing-aids/best-hearing-aids/
RNID Blog on Hearing Health Technology: Discusses innovations like AI-powered
hearing aids and direct-to-consumer models.
https://rnid.org.uk/2024/12/imagining-the-future-of-hearing-health-uks-national-technology-adviser-dr-dave-smith/
PMC Disclaimer on Medical Content: Highlights the importance of disclaimers in
health-related advertising.
https://pmc.ncbi.nlm.nih.gov/about/disclaimer/